Request for Demo?

Emvólio
The Portable Refrigerator
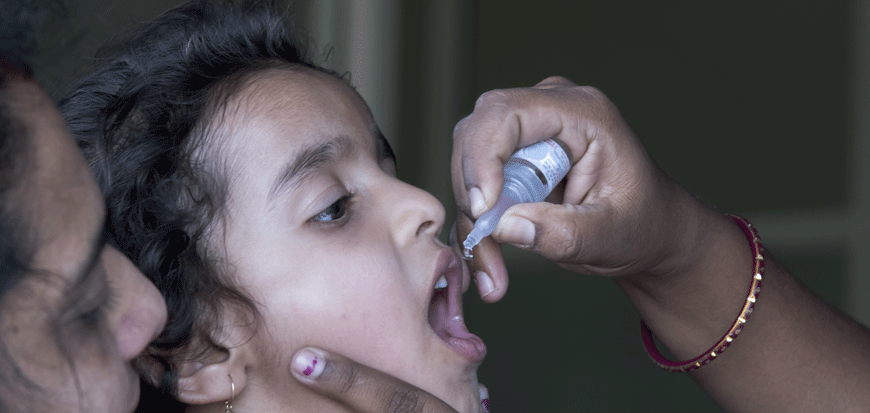
The team of engineers at Blackfrog have developed Emvólio - a portable active cooling (battery-powered) device that provides a platform for the delivery of vaccines and other biologicals like blood, serums, and viral cultures, which require strict temperature control between 2°C and 8°C for up to 12 hours in the field. The product is prequalified by the World Health Organisation (WHO PQS E003/134) and conforms to IEC 60601 standards.
Emvólio for Immunization
(Vaccine Carrier)
Issues with the Current Cold Chain System



Salient Features of Emvólio:
*12h battery life at test condition of 43°C ambient temperature and 5°C chamber temperature and 45 hours battery life at 25°C ambient temperature
| Cold-life usage duration* | 45+ hours |
| Capacity | 1.55 liters |
| Assured temperature maintenance | YES |
| Display (Feedback to user) | YES |
| Pre-cooling or conditioning required | NO |
| Online monitoring & report generation | YES |
| Portable & Rugged | YES |
| Leak Resistant | YES |

Emvólio for Biologicals
Emvólio for Consumer Applications
References
[1]: World Health Organization. (2005). Monitoring vaccine wastage at country level: guidelines for programme managers (No. WHO/V&B/03.18.Rev1). World Health Organization[2]: WHO-PQS Specifications for a portable refrigerator for transport of vaccines in the last- mile.
[3]: https://www.cdc.gov/breastfeeding/recommendations/handling_breastmilk.htm